Assign a systematic name to each of the following ions.
(c) [Fe(H2O)5NCS]2+
(d) [Cr(NH3)2(C2O4)2]-
 McMurry 8th Edition
McMurry 8th Edition Ch.21 - Transition Elements and Coordination Chemistry
Ch.21 - Transition Elements and Coordination Chemistry Problem 21.80a
Problem 21.80a Verified step by step guidance
Verified step by step guidance
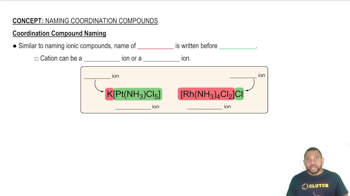


Assign a systematic name to each of the following ions.
(c) [Fe(H2O)5NCS]2+
(d) [Cr(NH3)2(C2O4)2]-
Based on the wavelength of maximum absorption of the cobalt complexes, arrange the following ligands in a spectrochemical series from weakest-field to strongest-field ligand.
(a) Cl- < NCS- < H2O < NH3
(b) Cl- < NCS- < H2O < NH3
(c) H2O < Cl- < NH3 < NCS-
(d) Cl- < H2O < NCS- < NH3
What is the systematic name for each of the following coordination compounds?
(c) [Co(NH3)4Br2]Br
(d) Cu(gly)2
Constitutional isomers of a ruthenium(II) coordination compound are shown below.
(a) Give the formula and name for structures 1-3.
(b) Which structures are linkage isomers?
(c) Which structures are ionization isomers?
Six isomers for a square planar palladium(II) complex that contains two Cl-and two SCN-ligands are shown below.
(a) Which structures are cis-trans isomers?
(b) Which structures are linkage isomers?
Which of the following complexes can exist as diastereoisomers?
(a) [Cr(NH3)2Cl4]-
(b) [Co(NH3)5Br]2+
(c) [MnCl2Br2]2- (tetrahedral)
(d) [Pt(NH3)2Br2]2-