Phosphorus is present in seawater to the extent of 0.07 ppm by mass. Assuming that the phosphorus is present as dihydrogenphosphate, H2PO4-, calculate the correspond-ing molar concentration of H2PO4- in seawater.
The Ogallala aquifer described in the Closer Look box in Section 18.3, provides 82% of the drinking water for the people who live in the region, although more than 75% of the water that is pumped from it is for irrigation. Irrigation withdrawals are approximately 18 billion gallons per day. (a) Assuming that 2% of the rainfall that falls on an area of 600,000 km2 recharges the aquifer, what average annual rainfall would be required to replace the water removed for irrigation?
 Verified step by step guidance
Verified step by step guidance
Verified Solution
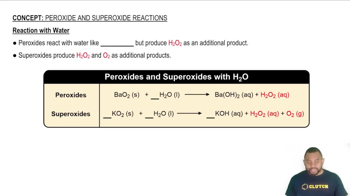
Key Concepts
Aquifer Recharge

Water Balance

Irrigation Water Use
The enthalpy of evaporation of water is 40.67 kJ/mol. Sunlight striking Earth's surface supplies 168 W per square meter (1 W = 1 watt = 1 J/s). (a) Assuming that evaporation of water is due only to energy input from the Sun, calculate how many grams of water could be evaporated from a 1.00 square meter patch of ocean over a 12-h day
The enthalpy of fusion of water is 6.01 kJ/mol. Sunlight striking Earth's surface supplies 168 W per square meter (1 W = 1 watt = 1 J/s). (b) The specific heat capacity of ice is 2.032 J/g°C. If the initial temperature of a 1.00 square emter patch of ice is -5.0°C, what is its final temperature after being in sunlight for 12 h, assuming no phase changes and assuming that sunlight penetration uniformly to a depth of 1.00 cm?
The organic anion
is found in most detergents. Assume that the anion under-goes aerobic decomposition in the following manner: C18H29SO3- + 51 O2 → 36 CO2(aq) + 28 H2O (l) + 2 H+(aq) + 2 SO42-(aq) What is the total mass of O2 required to biodegrade 10.0 g of this substance?
Magnesium ions are removed in water treatment by the addition of slaked lime, Ca(OH)2. Write a balanced chemical equation to describe what occurs in this process
In the lime soda process once used in large scale munici-pal water softening, calcium hydroxide prepared from lime and sodium carbonate are added to precipitate Ca2+ as CaCO3(s) and Mg2+ as Mg(OH)2(s): Ca2+(aq) + CO32-(aq) → CaCO3(s) Mg2+(aq) + 2 OH-(aq) → MgOH2(aq) How many moles of Ca(OH)2 and Na2CO3 should be added to soften (remove the Ca2+ and Mg2+) 1200 L of water in which [Ca2+] = 5.0x10-4 M and [Mg2+] = 7.0x10-4 M?
