Sulfur tetrafluoride (SF4) reacts slowly with O2 to form sulfur tetrafluoride monoxide (OSF4) according to the following unbalanced reaction: SF4(g) + O2(g) → OSF4(g) The O atom and the four F atoms in OSF4 are bonded to a central S atom. (b) Write a Lewis structure of OSF4 in which the formal charges of all atoms are zero.
Ch.9 - Molecular Geometry and Bonding Theories

Brown15th EditionChemistry: The Central ScienceISBN: 9780137542970Not the one you use?Change textbook
Chapter 9, Problem 113a
Methyl isocyanate, CH3NCO, was made infamous in 1984 when an accidental leakage of this compound from a storage tank in Bhopal, India, resulted in the deaths of about 3800 people and severe and lasting injury to many thousands more. (a) Draw a Lewis structure for methyl isocyanate.
 Verified step by step guidance
Verified step by step guidance1
Identify the total number of valence electrons in the molecule. Carbon (C) has 4, hydrogen (H) has 1, nitrogen (N) has 5, and oxygen (O) has 6. Calculate the total valence electrons for CH3NCO.
Determine the central atom. In this case, carbon (C) is often the central atom in organic molecules. Arrange the atoms in a reasonable structure: CH3 connected to NCO.
Draw single bonds between the central carbon and the surrounding atoms: C-H, C-N, and N-C-O. This uses up some of the valence electrons.
Distribute the remaining valence electrons to satisfy the octet rule for each atom, starting with the most electronegative atoms (O and N).
Adjust the structure to minimize formal charges, possibly by creating double or triple bonds, especially between C, N, and O, to ensure all atoms have a complete octet.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
2mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Lewis Structures
Lewis structures are diagrams that represent the bonding between atoms in a molecule and the lone pairs of electrons that may exist. They help visualize the arrangement of electrons and the connectivity of atoms, allowing chemists to predict molecular geometry and reactivity. In drawing a Lewis structure, one must account for the total number of valence electrons and ensure that each atom achieves a stable electron configuration, typically resembling that of noble gases.
Recommended video:
Guided course

Lewis Dot Structures: Ions
Valence Electrons
Valence electrons are the outermost electrons of an atom and are crucial in determining how an atom bonds with others. The number of valence electrons influences an atom's reactivity and the types of bonds it can form. For example, carbon has four valence electrons, allowing it to form four covalent bonds, while nitrogen has five, enabling it to form three bonds and hold a lone pair.
Recommended video:
Guided course
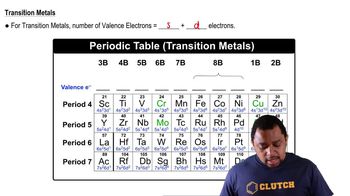
Transition Metals Valence Electrons
Molecular Geometry
Molecular geometry refers to the three-dimensional arrangement of atoms within a molecule. It is determined by the number of bonding pairs and lone pairs of electrons around the central atom, which affects the molecule's shape and properties. Understanding molecular geometry is essential for predicting the behavior of molecules in chemical reactions and their interactions with other substances.
Recommended video:
Guided course
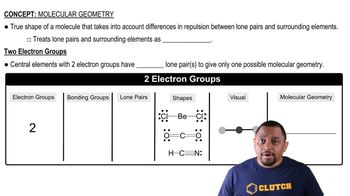
Molecular Geometry with Two Electron Groups
Related Practice
Textbook Question
400
views
Textbook Question
Sulfur tetrafluoride (SF4) reacts slowly with O2 to form sulfur tetrafluoride monoxide (OSF4) according to the following unbalanced reaction: SF4(g) + O2(g) → OSF4(g) The O atom and the four F atoms in OSF4 are bonded to a central S atom. (c) Use average bond enthalpies (Table 8.3) to estimate the enthalpy of the reaction. Is it endothermic or exothermic?
688
views
Textbook Question
Sulfur tetrafluoride 1SF42 reacts slowly with O2 to form sulfur
tetrafluoride monoxide 1OSF42 according to the following
unbalanced reaction:
SF41g2 + O21g2¡OSF41g2
The O atom and the four F atoms in OSF4 are bonded to a
central S atom.
(e) For each of the molecules you drew in part (d), state how many
fluorines are equatorial and how many are axial.
102
views
