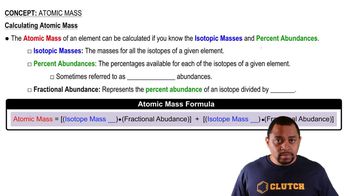
The atomic weight of boron is reported as 10.81, yet no atom of boron has the mass of 10.81 amu. Which is the best explanation?
a. The measurement of atomic mass is only reliable to two significant figures.
b. The atomic weight is an average of many individual atoms.
c. The atomic weight is an average of many isotopes of the same nuclear composition.