Using Lewis symbols and Lewis structures, make a sketch of the formation of NCl3 from N and Cl atoms, showing valence-shell electrons. (e) How many lone pairs of electrons are in the NCl3 molecule?

(b) How many bonding electrons are in the structure?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Bonding Electrons

Lewis Structures

Valence Electrons
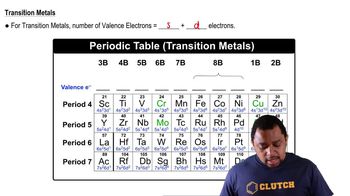
Using Lewis symbols and Lewis structures, diagram the formation of PF3 from P and F atoms, showing valence-shell electrons. (a) How many valence electrons does P have initially? (c) How many valence electrons surround the P in the PF3 molecule? (d) How many valence electrons surround each P in the PF3 molecule?
Using Lewis symbols and Lewis structures, diagram the formation of PF3 from P and F atoms, showing valence-shell electrons. (e) Does PF3 obey the octet rule?
(c) Would you expect the O—O bond in O2 to be shorter or longer than the O—O bond in compounds that contain an O—O single bond? Explain.
(a) Construct a Lewis structure for hydrogen peroxide, H2O2, in which each atom achieves an octet of electrons.
(c) Do you expect the O—O bond in H2O2 to be longer or shorter than the O—O bond in O2? Explain.
