(c) If 55.8 mL of a BaCl2 solution is needed to precipitate all the sulfate ion in a 752-mg sample of Na2SO4, what is the molarity of the BaCl2 solution?
A 4.36-g sample of an unknown alkali metal hydroxide is dissolved in 100.0 mL of water. An acid–base indicator is added, and the resulting solution is titrated with 2.50 M HCl(aq) solution. The indicator changes color, signaling that the equivalence point has been reached, after 17.0 mL of the hydrochloric acid solution has been added. (b) What is the identity of the alkali metal cation: Li+, Na+, K+, Rb+, or Cs+?

Verified Solution
Key Concepts
Titration and Equivalence Point

Molarity and Stoichiometry

Alkali Metal Hydroxides
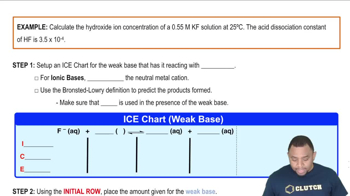
The distinctive odor of vinegar is due to acetic acid, CH3COOH, which reacts with sodium hydroxide according to: CH3COOH1aq2 + NaOH1aq2¡ H2O1l2 + NaCH3COO1aq2 If 3.45 mL of vinegar needs 42.5 mL of 0.115 M NaOH to reach the equivalence point in a titration, how many grams of acetic acid are in a 1.00-qt sample of this vinegar?
A 4.36-g sample of an unknown alkali metal hydroxide is dissolved in 100.0 mL of water. An acid–base indicator is added, and the resulting solution is titrated with 2.50 M HCl(aq) solution. The indicator changes color, signaling that the equivalence point has been reached, after 17.0 mL of the hydrochloric acid solution has been added. (a) What is the molar mass of the metal hydroxide?
An 8.65-g sample of an unknown group 2 metal hydroxide is dissolved in 85.0 mL of water. An acid–base indicator is added and the resulting solution is titrated with 2.50 M HCl(aq) solution. The indicator changes color, signaling that the equivalence point has been reached, after 56.9 mL of the hydrochloric acid solution has been added. (b) What is the identity of the metal cation: Ca2+, Sr2+, or Ba2+?
A solution of 100.0 mL of 0.200 M KOH is mixed with a solution of 200.0 mL of 0.150 M NiSO4. (a) Write the balanced chemical equation for the reaction that occurs.
A solution of 100.0 mL of 0.200 M KOH is mixed with a solution of 200.0 mL of 0.150 M NiSO4. (b) What precipitate forms?
