Fill in the blanks in the following table:
Cation Anion Formula Name
Lithium oxide
Fe2+ PO43-
Al2(SO4)3
Copper(II) nitrate
Cr3+ I−
MnClO2
Ammonium carbonate
Zinc perchlorate
Complete the first column of the table.
 Verified step by step guidance
Verified step by step guidance
Verified Solution
Key Concepts
Ionic Compounds

Nomenclature of Ions

Chemical Formulas
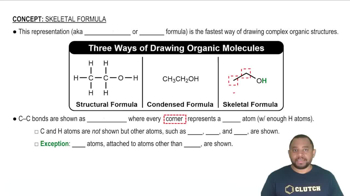
From the molecular structures shown here, identify the one that corresponds to each of the following species: (a) chlorine gas; (b) propane; (c) nitrate ion; (d) sulfur trioxide; (e) methyl chloride, CH3Cl.
Fill in the blanks in the following table:
Cation Anion Formula Name
Lithium oxide
Fe2+ PO43-
Al2(SO4)3
Copper(II) nitrate
Cr3+ I−
MnClO2
Ammonium carbonate
Zinc perchlorate
Complete the third column of the table.
Complete the fourth column of the table.
Cyclopropane is an interesting hydrocarbon. Instead of having three carbons in a row, the three carbons form a ring, as shown in this perspective drawing (see Figure 2.18 for a prior example of this kind of drawing):
Cyclopropane was at one time used as an anesthetic, but its use was discontinued, in part because it is highly flammable. (a) How does it differ from that of propane?
