Consider the following three reactions: (i) Ti(s) + 2 Cl2(g) → TiCl4(1g) (ii) C2H6(g) + 7 Cl2(g) → 2 CCl4(g) + 6 HCl(g) (iii) BaO(s) + CO2(g) → BaCO3(s) (c) For each of the reactions, predict the manner in which the change in free energy varies with an increase in temperature.

(b) Based on your general chemical knowledge, predict which of these reactions will have K>1. (i) 2 Mg(s) + O2 (g) ⇌ 2 MgO(s) (ii) 2 KI(s) ⇌ 2 K(g) + I2(g) (iii) Na2(g) ⇌ 2 Na(g) (iv) 2 V2O5(s) ⇌ 4 V(s) + 5 O2(g)
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
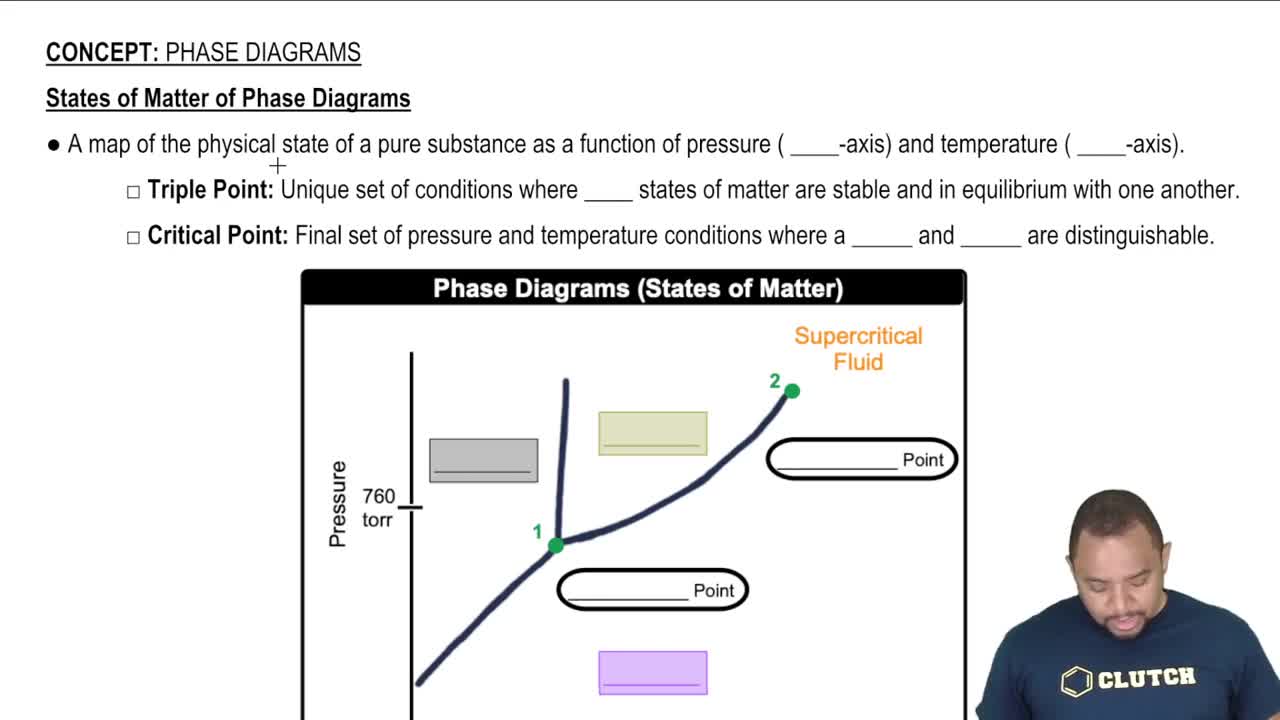
Key Concepts
Equilibrium Constant (K)

Gibbs Free Energy (ΔG)

Phase States and Their Influence on K
Using the data in Appendix C and given the pressures listed, calculate Kp and ΔG for each of the following reactions:
(a) N2(g) + 3 H2(g) → 2 NH3(g) PN2 = 2.6 atm, PH2 = 5.9 atm, PNH3 = 1.2 atm
(b) 2 N2H4(g) + 2 NO2(g) → 3 N2(g) + 4 H2O(g) PN2H4 = PNO2 = 5.0 × 10-2 atm, PN2 = 0.5 atm, PH2O = 0.3 atm
(c) N2H4(g) → N2(g) + 2 H2(g) PN2H4 = 0.5 atm, PN2 = 1.5 atm, PH2 = 2.5 atm
(a) For each of the following reactions, predict the sign of ΔH° and ΔS° without doing any calculations. (i) 2 Mg(s) + O2 (g) ⇌ 2 MgO(s) (ii) 2 KI(s) ⇌ 2 K(g) + I2(g) (iii) Na2(g) ⇌ 2 Na(g) (iv) 2 V2O5(s) ⇌ 4 V(s) + 5 O2(g)
(c) In each case, indicate whether K should increase or decrease with increasing temperature. (i) 2 Mg(s) + O2 (g) ⇌ 2 MgO(s) (ii) 2 KI(s) ⇌ 2 K(g) + I2(g) (iii) Na2(g) ⇌ 2 Na(g) (iv) 2 V2O5(s) ⇌ 4 V(s) + 5 O2(g)
The oxidation of glucose (C6H12O6) in body tissue produces CO2 and H2O. In contrast, anaerobic decomposition, which occurs during fermentation, produces ethanol (C2H5OH) and CO2.
(a) Using data given in Appendix C, compare the equilibrium constants for the following reactions:
C6H12O6(s) + 6 O2(g) ⇌ 6 CO2(g) + 6 H2O(l)
C6H12O6(s) ⇌ 2 C2H5OH(l) + 2 CO2(g)
