Textbook Question
Write the chemical equation and the Kb expression for the reaction of each of the following bases with water: (c) benzoate ion, C6H5CO2-
512
views
 Verified step by step guidance
Verified step by step guidance

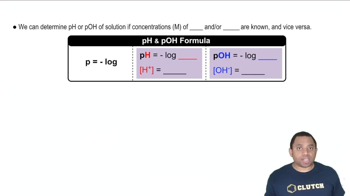

Write the chemical equation and the Kb expression for the reaction of each of the following bases with water: (c) benzoate ion, C6H5CO2-
Ephedrine, a central nervous system stimulant, is used in nasal sprays as a decongestant. This compound is a weak organic base: C10H15ON1aq2 + H2O1l2 Δ C10H15ONH+1aq2 + OH-1aq2 A 0.035 M solution of ephedrine has a pH of 11.33. (b) Calculate Kb for ephedrine.
Codeine 1C18H21NO32 is a weak organic base. A 5.0 * 10-3M solution of codeine has a pH of 9.95. Calculate the value of Kb for this substance. What is the pKb for this base?