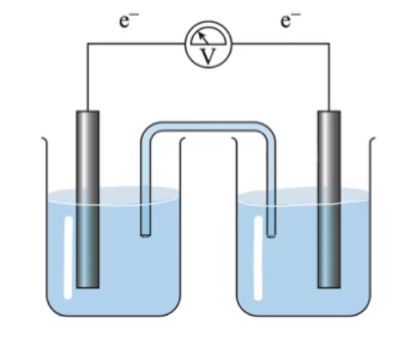
In this video we're going to take a look at the galvanic cell electrodes. Here we're going to say recall all electrochemical cells possess 2 half cell compartments, one for oxidation and the other for reduction. Remember the anode equals oxidation and a way to remember that is an Aux and then the cathode equals reduction. A way to remember that is red cat.
Now, before we start filling in the rest of this, let's just talk about some key things. We're going to say for a galvanic cell, in terms of its electrodes, the anode is negatively charged and the cathode is positively charged. What effect will this have on the electrodes over time? All right, so we know the anode is where oxidation occurs. Here we're looking at the zinc electrode much more closely and we're going to say that zinc is being oxidized to zinc on. Now what's happening here is electrons are literally leaving the electrode and traveling towards the cathode.
The cathode is positively charged, it's gaining electrons. Here copper 2 plus would be reduced to copper solid. Now what effect is this going to have? We're going to say here we see our zinc solid surface here and what's happening is we have electrons traveling up the electrode leaving the electrode to go to the cathode. What effect does this have? Well my Zinc is losing electrons. As a result of this it starts to produce Zinc 2 plus ions which become more and more dissolved within my solution. So electrons don't weigh very much. But overtime if you keep losing electrons those little masses do add up.
Eventually, you're going to lose a good chunk of this anime, and let's say that this is what's left. The rest of it's gone. You've been losing electrons consistently over time, so you're you're losing the size of your atom. So here we'd say that the anode is our negative electrode. It loses electrons and overtime it causes a decrease in mass. Here we say that the anode dissolves away. What's happening to the captain? Well, the cathode is our positive electrode, yes, but over time it's gaining more and more electrons. These electrons are depositing themselves onto the surface of the cathode.
It is in a solution of its own ions, in this case copper 2 plus ions. The surface is becoming more and more negatively charged with the electrons and gains, which is going to attract these positive ions floating around. They're going to move towards the surface. Two plus ion seeing electrons on the surface connects to them. Negative charge, positive charge neutralize each other. So the copper ions are going to start to coat the surface of this electrode. So over time, the cathode is going to get bulkier, it's going to get bigger, it's gaining mass. We're going to say the cathode gains electrons, causing the surface to become more negatively charged, attracting cations to it. And this over time causes an increase in mass.
Here we would say that the cathode plates out, one gets skinnier, 1 gets bigger. Now here we talked about our salt bridge in earlier videos. Remember a salt bridge there just has neutral ions within it. So we know that the negative ions here would go towards the anode compartment and positive ions go here towards the Catholic apartment. But why exactly are these negative ions going towards the anode side? Well, what's happening? More and more positive ions are coming off of the anode, so your solutions can become saturated with these positive ions. So much so that if too many of them are hanging around, the electrolyte solution is super positive. These negative electrons won't want to leave.
They'll be like why should I leave if my solution has all these dissolved positive ions around opposite charges attract. That's why we need a salt bridge. The salt bridge has these negative ions go into the solution, neutralizing these positive cations that are being produced, keeping their concentration down, and in that way the electrons are not conflicted. Do I go towards the cathode, which is becoming less positive over time as it gains more and more electrons on its surface? Or should I stick around and hang out with these dissolved cat ions in my solution? Here electrons don't have to worry about that because again, the negative ions within the salt bridge are neutralizing. These excess cations are being produced in the anode chamber.
Now here if we're talking about reactions, remember zinc is being oxidized to zinc. So what would that look like? We'd have zinc solid benoxidized to zinc 2 plus aqueous and the two electrons that are lost in the Catholic compartment, copper 2 plus is reduced, which means it gains 2 electrons to produce copper solid. We can't slot intermediates, which in this case are the electrons, and what's left behind gives us our overall reaction. So here we have zinc 2 plus or zinc solid plus. Copper 2 plus gives me zinc 2 plus plus copper solid. So this would be the overall reaction that occurs from this electrochemical cell dealing with our zinc electrode and copper electrodes.
Zn
2
+
+
Cu
2
+
→
Zn
2
+
+
Cu
2
+