9. Quantum Mechanics
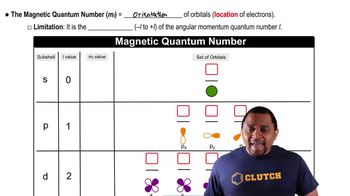
Quantum Numbers: Magnetic Quantum Number
Problem 58
Textbook Question
Textbook QuestionGive the values for n, l, and ml for (a) each orbital in the 3p subshell.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
673
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos