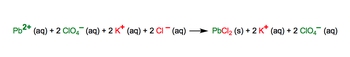
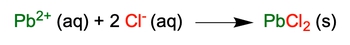In chemistry, understanding the transition from a molecular equation to a complete ionic equation is crucial for analyzing reactions in aqueous solutions. A molecular equation presents all reactants and products in their molecular form, including solids, liquids, and gases, while a complete ionic equation breaks down only the aqueous compounds into their constituent ions.
When dealing with complete ionic equations, it is essential to recognize that only aqueous compounds dissociate into ions. Solids, liquids, and gases remain intact and do not break apart. To determine whether a compound is aqueous, solubility rules are applied, which help identify which substances will dissolve in water and form ions.
To derive a complete ionic equation from a molecular equation, one must distribute the coefficients of each compound correctly. This ensures that the number of ions represented reflects the stoichiometry of the reaction. For example, if a compound has a coefficient of 2, it will produce twice the number of ions in the complete ionic equation.
In summary, the complete ionic equation provides a clearer picture of the actual species present in solution during a chemical reaction, allowing for a better understanding of the interactions between ions. This approach is fundamental in predicting the outcomes of reactions and understanding the behavior of different compounds in aqueous environments.