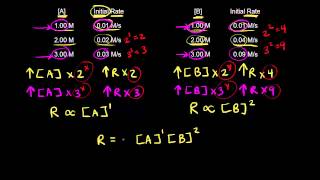15. Chemical Kinetics
Rate Law
Problem 140
Textbook Question
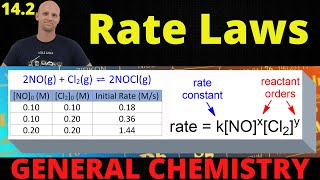
Textbook QuestionThe rate constant for the first-order decomposition of gaseous N2O5 to NO2 and O2 is 1.7 * 10-3 s-1 at 55 °C. (b) Use the data in Appendix B to calculate the initial rate at which the reaction mixture absorbs heat (in J/s). You may assume that the heat of the reaction is independent of temperature.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
496
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos