18. Aqueous Equilibrium
Titrations: Strong Acid-Strong Base
Problem 68
Textbook Question
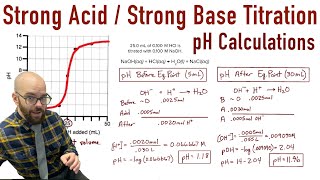
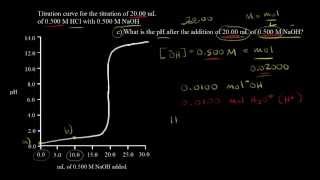
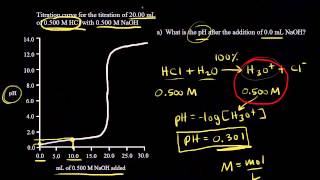
Textbook QuestionA 20.0-mL sample of 0.125 M HNO3 is titrated with 0.150 M NaOH. Calculate the pH for at least five different points throughout the titration curve and sketch the curve. Indicate the volume at the equivalence point on your graph.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
16mPlay a video:
401
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos