13. Liquids, Solids & Intermolecular Forces
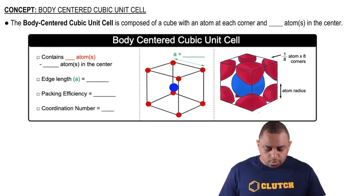
Body Centered Cubic Unit Cell
Problem 48
Textbook Question
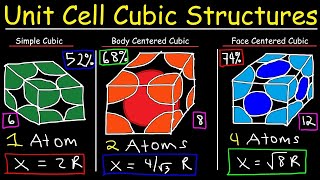
Textbook QuestionIron crystallizes in a body-centered cubic unit cell with an edge length of 287 pm. Iron metal has a density of 7.86 g>cm3 and a molar mass of 55.85 g. Calculate a value for Avogadro's number.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1960
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos