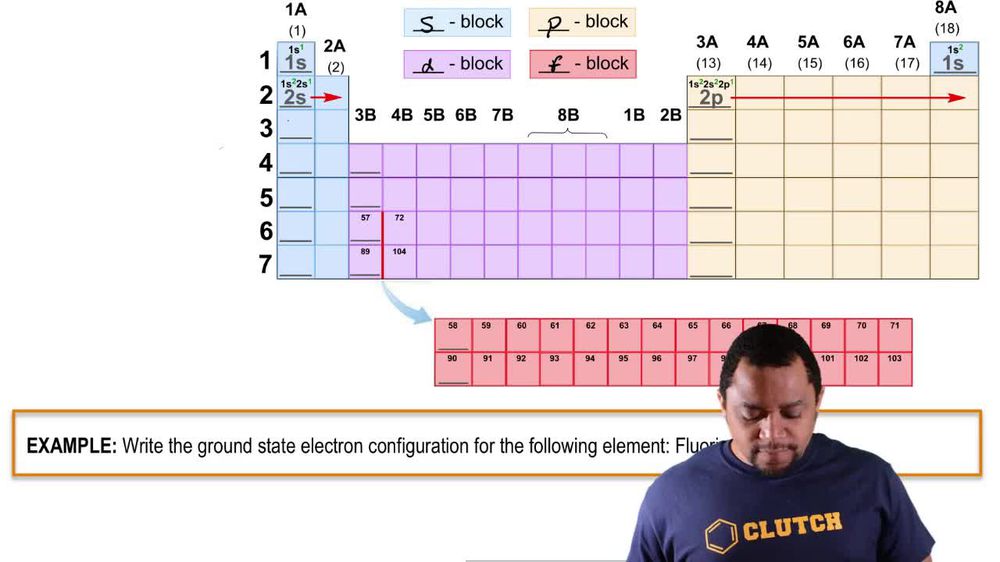
Periodic Trends
Periodic trends refer to predictable patterns in elemental properties across the periodic table, such as electronegativity, ionization energy, and electron affinity. As you move from left to right across a period, elements generally become more electronegative and have more negative electron affinities. This trend explains why group 7A elements, with their higher effective nuclear charge and electron configurations, have a stronger attraction for additional electrons compared to group 6A elements.