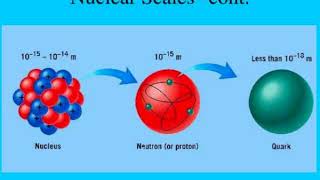
21. Nuclear Chemistry
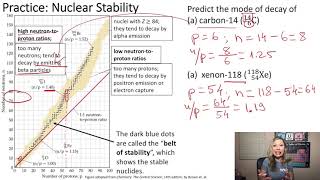
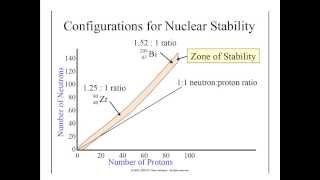
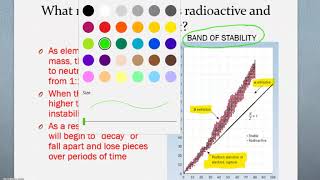
Neutron to Proton Ratio
Problem 77
Textbook Question
Textbook QuestionIn 2010, a team of scientists from Russia and the United States reported creation of the first atom of element 117, which is named tennessine, and whose symbol is Ts. The synthesis involved the collision of a target of 24997Bk with accelerated ions of an isotope which we will denote Q. The product atom, which we will call Z, immediately releases neutrons and forms 294117Ts: 24997Bk + Q¡Z¡294117Ts + 3 10n (b) Isotope Q is unusual in that it is very long-lived (its half-life is on the order of 1019 yr) in spite of having an unfavorable neutron-to-proton ratio (Figure 21.1). Can you propose a reason for its unusual stability?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
400
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos