18. Aqueous Equilibrium
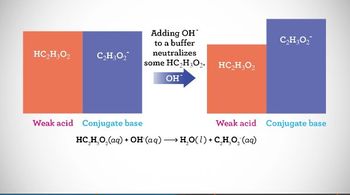
Intro to Buffers
Problem 88
Textbook Question
Textbook QuestionTwo buffers are prepared by adding an equal number of moles of formic acid (HCOOH) and sodium formate (HCOONa) to enough water to make 1.00 L of solution. Buffer A is prepared using 1.00 mol each of formic acid and sodium formate. Buffer B is prepared by using 0.010 mol of each. (b) Which buffer will have the greater buffer capacity?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1312
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos