24. Transition Metals and Coordination Compounds
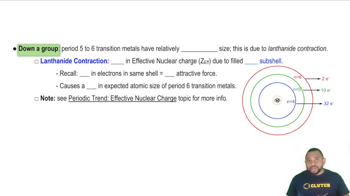
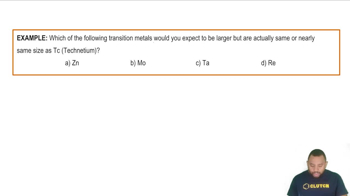
Atomic Radius & Density of Transition Metals
Problem 21.128a
Textbook Question
Textbook QuestionThere are two possible [M(OH)4]- complexes of first-series transition metals that have three unpaired electrons.
(a) What are the oxidation state and the identity of M in these complexes?
(b) Using orbital diagrams, give a valence bond description of the bonding in each complex.
(c) Based on common oxidation states of first-series transition metals (Figure 21.6), which [M(OH)4]- complex is more likely to exist?
<QUESTION REFERENCES FIGURE 21.6>-
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
100
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos







![13.1 Transition Metal Properties Overview [HL IB Chemistry]](https://img.youtube.com/vi/OAC8rToqfc4/mqdefault.jpg)

