16. Chemical Equilibrium
ICE Charts
Problem 161
Textbook Question
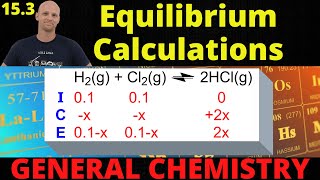
Textbook QuestionThe equilibrium constant for the dimerization of acetic acid in benzene solution is 1.51 * 102 at 25 °C. 2 CH3CO2H ∆ 1CH3CO2H22 Kc = 1.51 * 102 at 25 °C (b) What is the osmotic pressure of the solution at 25 °C?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
13mPlay a video:
882
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos