17. Acid and Base Equilibrium
Triprotic Acids and Bases Calculations
Problem 152
Textbook Question
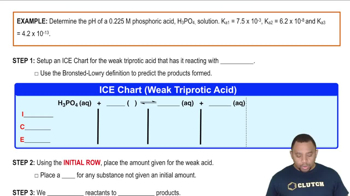
Textbook QuestionA 7.0 mass % solution of H3PO4 in water has a density of 1.0353 g/mL. Calculate the pH and the molar concentrations of all species present (H3PO4, H2PO4-, PO43-, H3O+ , and OH-) in the solution. Values of equilibrium constants are listed in Appendix C.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
16mPlay a video:
510
views
1
rank
Was this helpful?
Related Videos
Related Practice