13. Liquids, Solids & Intermolecular Forces
Heating and Cooling Curves
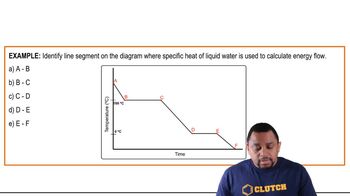
Problem 68
Textbook Question
Textbook QuestionCalculate the amount of heat required to completely sublime 50.0 g of solid dry ice (CO2) at its sublimation temperature. The heat of sublimation for carbon dioxide is 32.3 kJ/mol.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
5781
views
4
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos