13. Liquids, Solids & Intermolecular Forces
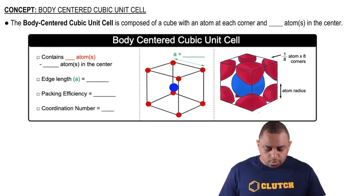
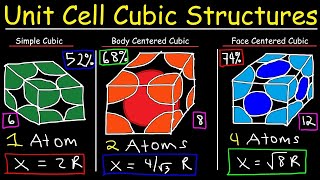
Body Centered Cubic Unit Cell
Problem 34a
Textbook Question
Textbook QuestionSodium metal (atomic weight 22.99 g>mol) adopts a body-centered cubic structure with a density of 0.97 g>cm3. (b) If sodium didn't react so vigorously, it could float on water. Use the answer from part (a) to estimate the density of Na if its structure were that of a cubic close-packed metal. Would it still float on water?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
1687
views
1
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos