Textbook Question
Write a Lewis structure that obeys the octet rule for each molecule or ion. Include resonance structures if necessary and assign formal charges to each atom. b. CO32– c. ClO– d. NO2–
1
views

 Verified step by step guidance
Verified step by step guidance

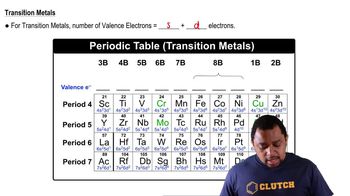
Write a Lewis structure that obeys the octet rule for each molecule or ion. Include resonance structures if necessary and assign formal charges to each atom. b. CO32– c. ClO– d. NO2–
Write the Lewis structure for each molecule or ion. c. BrO-
Write a Lewis structure that obeys the octet rule for each molecule or ion. Include resonance structures if necessary and assign formal charges to each atom. a. SeO2
Write the Lewis structure for each molecule or ion. b. OH-
Write the Lewis structure for each molecule or ion. a. N2H2 b. N2H4 c. C2H2
Write the Lewis structure for each molecule or ion. a. H3COCH3