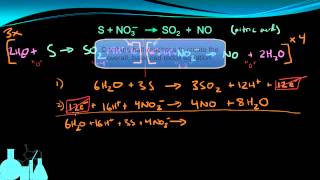6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Acidic Solutions
Problem 97
Textbook Question
Textbook QuestionA disproportionation reaction is an oxidation–reduction reaction in which the same substance is oxidized and reduced. Complete and balance the following disproportionation reactions: (b) MnO42-1aq2 ¡ MnO4-1aq2 + MnO21s2 (acidic solution)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
1093
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos