19. Chemical Thermodynamics
Entropy
Multiple Choice
Multiple ChoicePredict the sign of ∆S in the system for each of the following processes:
a) Ag+ (aq) + Br - (aq) → AgBr (s)
b) CI2 (g) → 2 CI - (g)
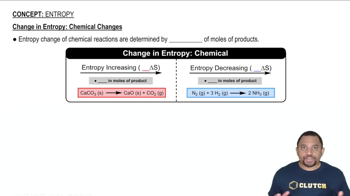
c) CaCO3 (s) → CaO (s) + CO2 (g)
d) Pb (s) at 50°C → Pb (s) at 70°C
A
a) ∆S = + b) ∆S = + c) ∆S = + d) ∆S = +
B
a) ∆S = - b) ∆S = + c) ∆S = - d) ∆S = +
C
a) ∆S = - b) ∆S = + c) ∆S = - d) ∆S = -
D
a) ∆S = - b) ∆S = + c) ∆S = + d) ∆S = +
2112
views
4
rank
3
comments
Related Videos
Related Practice
Showing 1 of 16 videos