10. Periodic Properties of the Elements
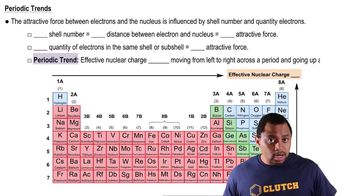
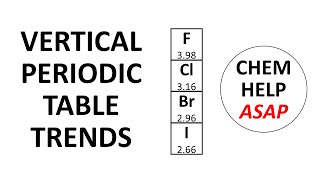
Periodic Trend: Effective Nuclear Charge
Open Question
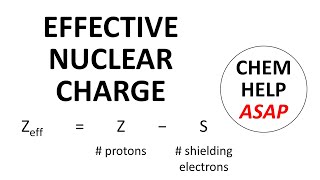
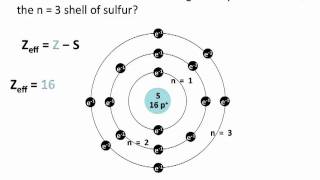
Open QuestionThe shielding of electrons gives rise to an effective nuclear charge, Zeff, which explains why boron is larger than oxygen. Estimate the approximate Zeff felt by a valence electron of boron and oxygen, respectively?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
179
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos