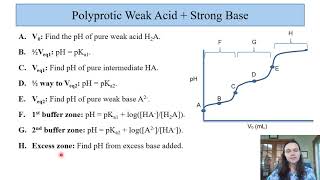
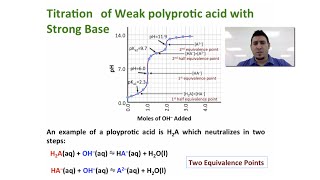
18. Aqueous Equilibrium
Titrations: Diprotic & Polyprotic Buffers
Problem 132
Textbook Question
Textbook QuestionA 0.867-g sample of an unknown acid requires 32.2 mL of a 0.182 M barium hydroxide solution for neutralization. Assuming the acid is diprotic, calculate the molar mass of the acid.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1153
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos