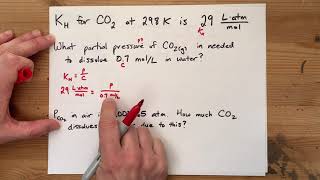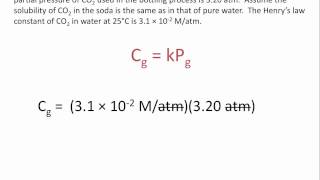14. Solutions
Henry's Law Calculations
Problem 84
Textbook Question
Textbook QuestionFish generally need an O2 concentration in water of at least 4 mg/L for survival. What partial pressure of oxygen above the water in atmospheres at 0 °C is needed to obtain this concentration? The solubility of O2 in water at 0 °C and 1 atm partial pressure is 2.21 * 10-3 mol>L.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
832
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos