12. Molecular Shapes & Valence Bond Theory
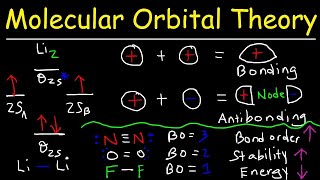
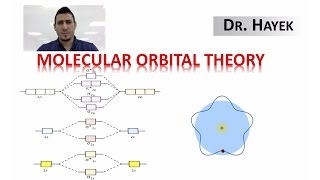
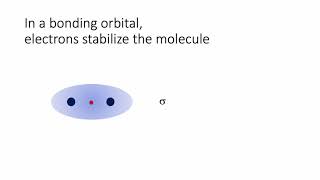
Molecular Orbital Theory
Problem 115
Textbook Question
Textbook QuestionCyclooctatetraene dianion, C8H8 2-, is an organic ion with the structure shown. Considering only the p bonds and not the s bonds, cyclooctatetraene dianion can be described by the following energy diagrams of its p molecular orbitals:

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
809
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos