8. Thermochemistry
Hess's Law
Problem 38b
Textbook Question
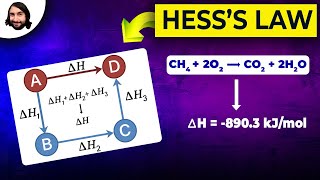
Textbook QuestionThe reaction of A with B to give D proceeds in two steps: (1) A + B → C ΔH° = -20 kJ (2) C + B → D ΔH° = +50 kJ (3) A + 2B → D ΔH° = ? (a) Which Hess's law diagram represents the reaction steps and the overall reaction? Diagram 1 Diagram 2
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
511
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos