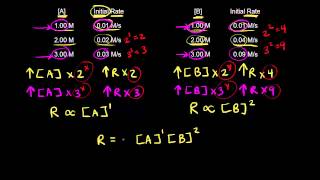15. Chemical Kinetics
Rate Law
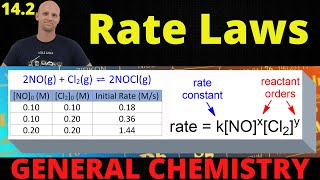
Problem 28a
Textbook Question
Textbook QuestionConsider a hypothetical reaction between A, B, and C that is first order in A, zero order in B, and second order in C. (e) By what factor does the rate change when the concentrations of all three reactants are tripled?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
926
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos