18. Aqueous Equilibrium
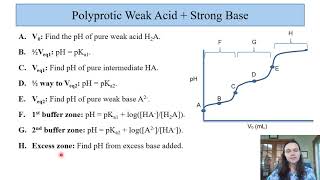
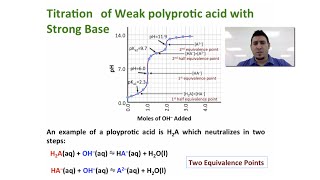
Titrations: Diprotic & Polyprotic Buffers
Problem 97a
Textbook Question
Textbook QuestionConsider the titration of 50.0 mL of 1.00 M H3PO4 with 1.00 M KOH. Calculate the pH after the addition of each of the following volumes of base. (a) 25.0 mL
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
214
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos