19. Chemical Thermodynamics
Entropy
Problem 23
Textbook Question
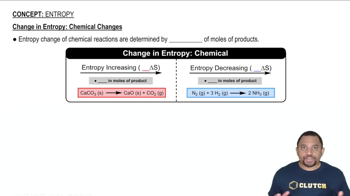
Textbook QuestionThe normal boiling point of Br21l2 is 58.8 °C, and its molar enthalpy of vaporization is ΔHvap = 29.6 kJ>mol. (a) When Br21l2 boils at its normal boiling point, does its entropy increase or decrease?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
827
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos