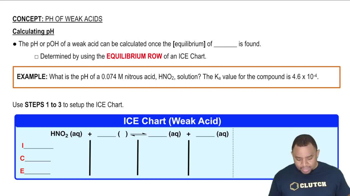
17. Acid and Base Equilibrium
pH of Weak Acids
Problem 26
Textbook Question
Textbook QuestionYou are asked to prepare a pH = 4.00 buffer starting from 1.50 L of 0.0200 M solution of benzoic acid 1C6H5COOH2 and any amount you need of sodium benzoate 1C6H5COONa2. (a) What is the pH of the benzoic acid solution prior to adding sodium benzoate?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
276
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos