17. Acid and Base Equilibrium
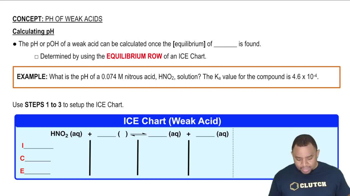
pH of Weak Acids
Problem 99
Textbook Question
Textbook QuestionA typical aspirin tablet contains 324 mg of aspirin (acetylsalicylic acid, C9H8O4), a monoprotic acid having Ka = 3.0 * 10-4. If you dissolve two aspirin tablets in a 300 mL glass of water, what is the pH of the solution and the percent dissociation?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
1560
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos