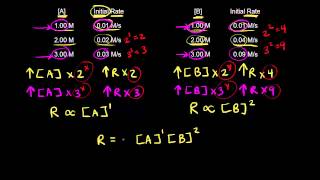15. Chemical Kinetics
Rate Law
Problem 127c
Textbook Question
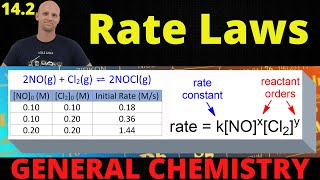
Textbook QuestionConsider the reversible, first-order interconversion of two molecules A and B: A kfkrBwhere kf = 3.0 * 10-3 s-1 is the rate constant for the forward reaction and kr = 1.0 * 10-3 s-1 is the rate constant for the reverse reaction. We'll see in Chapter 15 that a reaction does not go to completion but instead reaches a state of equilibrium with comparable concentrations of reactants and products if the rate constants kf and kr have comparable values. (a) What are the rate laws for the forward and reverse reactions?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
447
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos