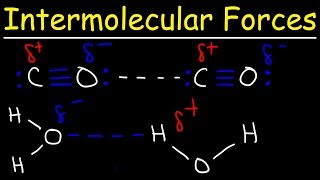
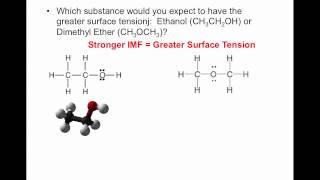
13. Liquids, Solids & Intermolecular Forces
Intermolecular Forces and Physical Properties
Problem 90
Textbook Question
Textbook QuestionIn Table 11.3, we saw that the viscosity of a series of hydro- carbons increased with molecular weight, doubling from the six-carbon molecule to the ten-carbon molecule.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
463
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos