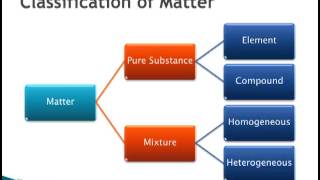
1. Intro to General Chemistry
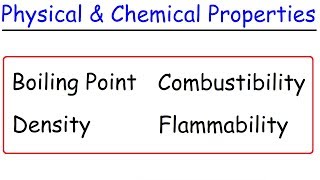
Physical Properties
Problem 19
Textbook Question
Textbook QuestionIn the process of attempting to characterize a substance, a chemist makes the following observations: The substance is a silvery white, lustrous metal. It melts at 649 C and boils at 1105 C. Its density at 20 C is 1.738 g/cm3. The substance burns in air, producing an intense white light. It reacts with chlorine to give a brittle white solid. The substance can be pounded into thin sheets or drawn into wires. It is a good conductor of electricity. Which of these characteristics are physical properties, and which are chemical properties?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
946
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos