20. Electrochemistry
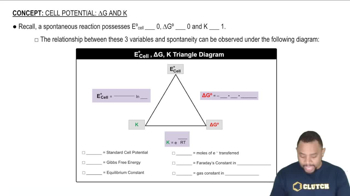
Cell Potential: ∆G and K
Problem 52a
Textbook Question
Textbook QuestionFor each of the following reactions, write a balanced equation, calculate the standard emf, calculate ∆G° at 298 K, and calculate the equilibrium constant K at 298 K. (c) In basic solution, Cr1OH231s2 is oxidized to CrO42-1aq2 by ClO-1aq2.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
16mPlay a video:
306
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos