9. Quantum Mechanics
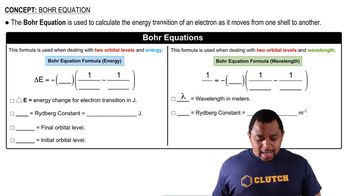
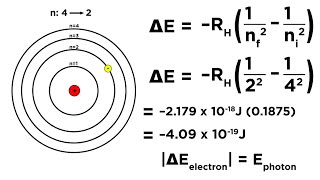
Bohr Equation
Problem 41b
Textbook Question
Textbook QuestionThe visible emission lines observed by Balmer all involved nf = 2. (b) Calculate the wavelengths of the first three lines in the Balmer series—those for which ni = 3, 4, and 5—and identify these lines in the emission spectrum shown in Figure 6.11.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
1272
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos