13. Liquids, Solids & Intermolecular Forces
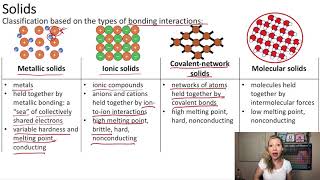
Atomic, Ionic, and Molecular Solids
Problem 62
Textbook Question
Textbook QuestionThe melting point of sodium metal is 97.8 °C, and the melting point of sodium chloride is 801 °C. What can you infer about the relative strength of metallic and ionic bonding from these melting points?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1900
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos