19. Chemical Thermodynamics
Entropy
Problem 50
Textbook Question
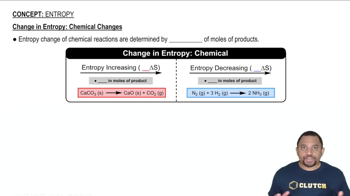
Textbook QuestionThree of the forms of elemental carbon are graphite, diamond, and buckminsterfullerene. The entropies at 298 K for graphite and diamond are listed in Appendix C. (b) What would you expect for the S° value of buckminsterfullerene (Figure 12.49, p. 509) relative to the values for graphite and diamond? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
849
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos