16. Chemical Equilibrium
ICE Charts
Problem 115
Textbook Question
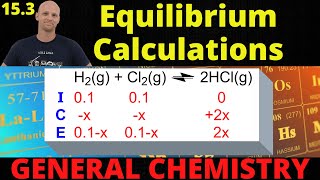
Textbook QuestionThe reaction of fumarate with water to form L-malate is catalyzed by the enzyme fumarase; Kc = 3.3 at 37°C. When a reaction mixture with [fumarate] = 1.56 * 10-3 M and [l -malate] = 2.27 * 10-3 M comes to equilibrium in the presence of fumarase at 37 °C, what are the equilibrium concentrations of fumarate and L-malate? (Water can be omit- ted from the equilibrium equation because its concentration in dilute solutions is essentially the same as that in pure water.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
827
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos