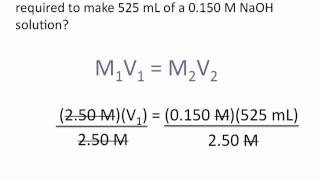6. Chemical Quantities & Aqueous Reactions
Dilutions
Problem 74a
Textbook Question
Textbook Question(b) If you dilute 10.0 mL of the stock solution to a final volume of 0.250 L, what will be the concentration of the diluted solution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
918
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos