Predict the stronger acid in each pair: (c) HBrO3 or HBrO2

Based on their compositions and structures and on conjugate acid–base relationships, select the stronger base in each of the following pairs: (b) BrO- or BrO2-
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
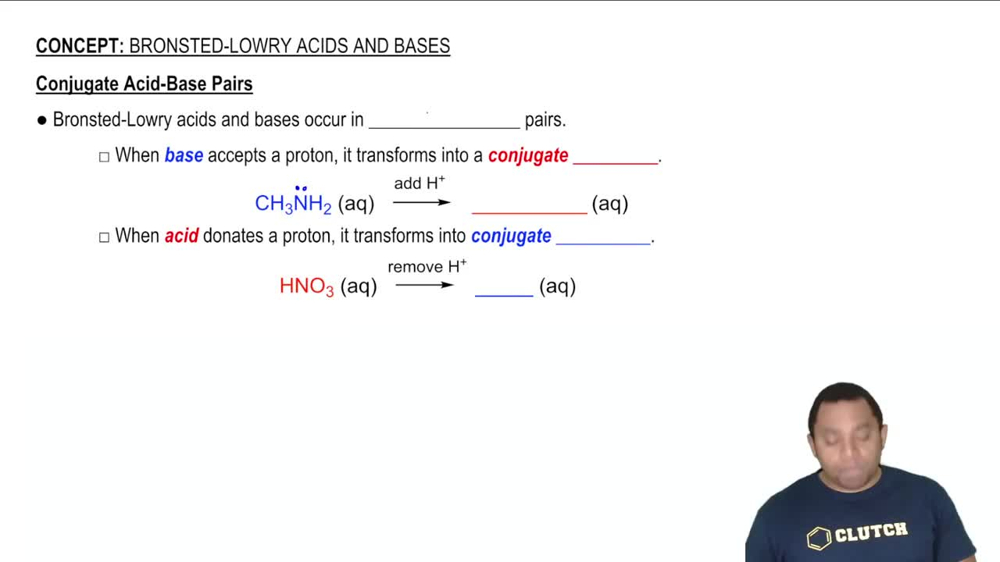
Conjugate Acid-Base Pairs
Oxidation States and Stability
Electronegativity and Basicity

Predict the stronger acid in each pair: (e) benzoic acid (C6H5COOH) or phenol (C6H5OH).
Based on their compositions and structures and on conjugate acid–base relationships, select the stronger base in each of the following pairs: (b) PO43- or AsO43-
Indicate whether each of the following statements is true or false. For each statement that is false, correct the statement to make it true. (a) In general, the acidity of binary acids increases from left to right in a given row of the periodic table. (b) In a series of acids that have the same central atom, acid strength increases with the number of hydrogen atoms bonded to the central atom. (c) Hydrotelluric acid 1H2Te2 is a stronger acid than H2S because Te is more electronegative than S.
Indicate whether each of the following statements is true or false. For each statement that is false, correct the statement to make it true. (a) Acid strength in a series of H¬A molecules increases with increasing size of A. (b) For acids of the same general structure but differing electronegativities of the central atoms, acid strength decreases with increasing electronegativity of the central atom. (c) The strongest acid known is HF because fluorine is the most electronegative element.
