17. Acid and Base Equilibrium
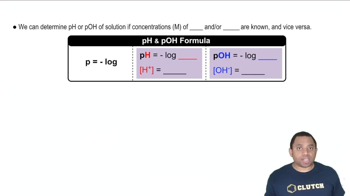
The pH Scale
Multiple Choice
Multiple ChoiceWhich of the following statements about aqueous solutions is/are true?
A
For an basic solution the concentration of H3O+ is greater than the concentration of OH-.
B
The pH of a neutral aqueous solution is 7.00 at all temperatures.
C
An acidic solution under normal conditions has a pH value less than 7.00.
D
If the concentration of H3O+ decreases then the concentration of OH- will also decrease.
E
The pH of aqueous solutions is less than 7.
1462
views
1
rank
Related Videos
Related Practice
Showing 1 of 10 videos