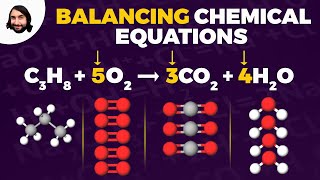
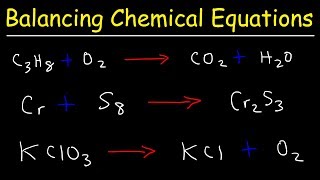
3. Chemical Reactions
Balancing Chemical Equations
Problem 151
Textbook Question
Textbook QuestionElement M is prepared industrially by a two-step procedure according to the following (unbalanced) equations: Assume that 0.855 g of M2O3 is submitted to the reaction sequence. When the HCl produced in step (2) is dissolved in water and titrated with 0.511 M NaOH, 144.2 mL of the NaOH solution is required to neutralize the HCl. (a) Balance both equations.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
449
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos