13. Liquids, Solids & Intermolecular Forces
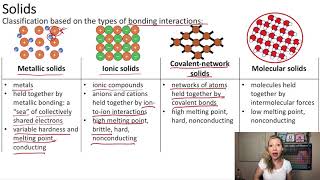
Atomic, Ionic, and Molecular Solids
Problem 44
Textbook Question
Textbook QuestionWhich solid in each pair has the higher melting point and why? d. H2O(s) or H2S(s)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
802
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos