17. Acid and Base Equilibrium
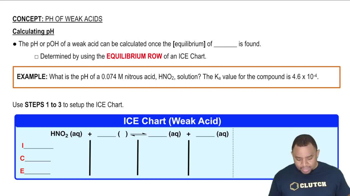
pH of Weak Acids
Problem 94
Textbook Question
Textbook QuestionA vitamin C tablet containing 250 mg of ascorbic acid 1C6H8O6; Ka = 8.0 * 10-52 is dissolved in a 250 mL glass of water. What is the pH of the solution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
15mPlay a video:
1319
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos